Process
Workflow


01.
This initial step involves registering the details of the quality control order, including product specifications, quantity, and any specific requirements.
02.
Once the order is registered, the next step involves creating Standard Operating Procedures (SOPs) tailored to the specific product or industry. These SOPs outline the standardized inspection guidelines, criteria, and parameters that will be followed during the quality inspection process.
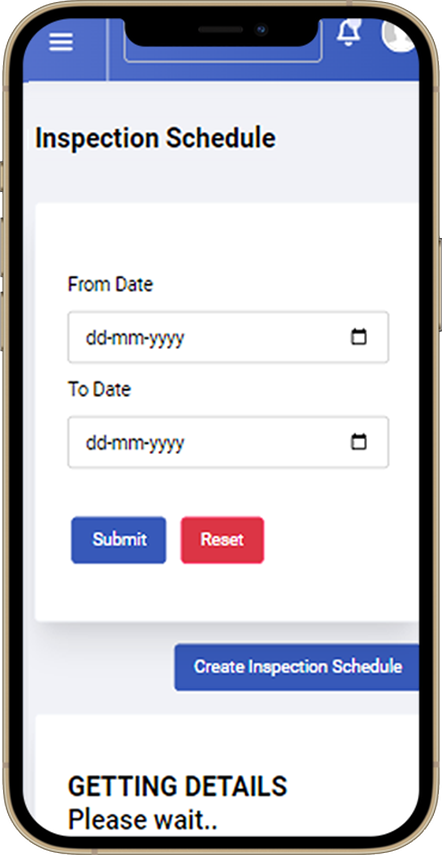
03.
The inspection is scheduled based on the availability of resources and the agreed timeline. This step involves coordinating with the inspection team, ensuring the necessary equipment and personnel are allocated for the inspection at the designated time.
04.
Upon completion of the inspection, a comprehensive report is generated, documenting the inspection findings, including any defects or anomalies detected. The report provides a detailed analysis of the quality assessment, allowing informed decisions regarding product acceptance, rejection, or corrective actions.
